Articles by Hannah Slater
Nivolumab was previously granted accelerated approval by the FDA for the treatment of patients with small cell lung cancer (SCLC) whose disease had progressed after platinum-based chemotherapy and at least 1 other line of therapy, but phase 3 trial results led to a decision to withdraw the indication.

Full approval has been granted by the FDA to osimertinib (Tagrisso) for use as adjuvant therapy following tumor resection among patients with non–small cell lung cancer whose tumors have EGFR exon 19 deletions or exon 21 L858R mutations.

The study evaluated lurbinectedin (Zepzelca) in combination with doxorubicin versus physician's choice of topotecan or cyclophosphamide/doxorubicin/vincristine for adult patients with small cell lung cancer whose disease progressed following 1 prior platinum-containing line of therapy.
These study results suggest that osimertinib as adjuvant therapy is an effective treatment strategy for patients with stage IB to IIIA EGFR-positive non-small cell lung cancer following complete tumor resection.

The FDA approved a 1500 mg fixed dose of durvalumab (Imfinzi) administered every 4 weeks for the treatment of unresectable stage III non-small cell lung cancer after chemoradiation therapy and previously treated advanced bladder cancer.
This study found that found that mini-strokes and heart attacks were significantly less common among patients with locally advanced non-small cell lung cancer who underwent proton therapy versus conventional photon-based radiation therapy.

Researchers identified molecular and cellular characteristics of anti-CD19 CAR T-cell infusion products associated with how patients with large B-cell lymphoma respond to treatment and experience adverse events.

The FDA granted breakthrough therapy designation to IMGN632 for the treatment of patients with relapsed or refractory blastic plasymacytoid dendritic cell neoplasm.

The FDA granted fast track designation to the novel “switchable” CAR-T cell therapy known as CLBR001 + SWI019 for the treatment of B-cell malignancies.

The FDA accepted an investigational new drug application providing clearance to begin an open-label, single-arm phase 1 clinical trial of ATA2271, a next-generation mesothelin targeting CAR T-cell therapy.

The study is designed to evaluate the safety and tolerability, pharmacodynamics, and preliminary efficacy of ACE1702 in patients with advanced or metastatic HER2-expressing solid tumors.

The FDA granted breakthrough therapy designation to osimertinib for the adjuvant treatment of patients with early-stage epidermal growth factor receptor-mutated non-small cell lung cancer after complete tumor resection with curative intent.

The FDA granted breakthrough therapy designation to MK-6482 for the treatment of patients with von Hippel-Lindau disease-associated renal cell carcinoma and orphan drug designation to MK-6482 for von Hippel-Lindau disease.

The agent is now the first and only approved CAR T-cell therapy to treat adult patients with relapsed or refractory mantle cell lymphoma.
The National Comprehensive Cancer Network published a document intended for patients to understand how CAR-T cells work and what side effects are associated with the treatment.
Patients with chemotherapy-naïve, locally advanced, or metastatic non-small cell lung cancer who were treated with tiragolumab plus an anti-PD-L1 agent showed better efficacy versus single-agent checkpoint inhibitor therapy alone.

The FDA approved oral selinexor for the treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma after at least 2 lines of systemic therapy.
Researchers found that a high dose of CAR T-19 may be more effective than a low dose at inducing a complete response without excessive toxicity.
Mount Sinai Health System announced that they will be using the allogeneic stem cell therapy remestemcel-L in patients with COVID-19, and have already tested the therapy in 10 patients.

Researchers found that the potential availability of CAR T-cell therapies for large B-cell lymphomas with lower adverse event rates that are suitable for outpatient administration may reduce the total costs of care.

The FDA cleared the investigational new drug (IND) application for the use of CYNK-001 in adults with COVID-19.

The planned trial will investigate the safety and efficacy of the novel multi-tumor associated antigen T-cell therapy in patients with post-transplant acute myeloid leukemia.

The biologic license application is supported by data from the phase II ZUMA-2 trial, which is currently assessing the CAR T-cell therapy for the treatment of adult patients with relapsed or refractory mantle cell lymphoma.
Patients with either relapsed or refractory non-Hodgkin lymphoma or chronic lymphocytic leukemia treated with CAR NK cells had a response without the development of cytokine release syndrome, neurotoxicity, or graft-versus-host disease.
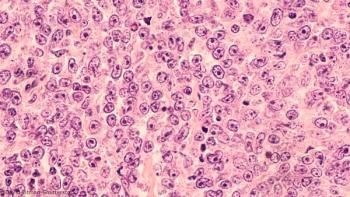
The data was presented at the EHA-EBMT 2nd European CAR T-Cell Meeting, which took place on January 30, 2020 in Barcelona, Spain, and showed encouraging signs of a manageable safety profile in adults with relapsed/refractory DLBCL.
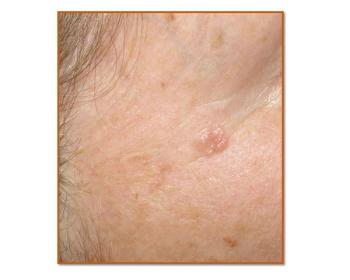
The recommendations defined when and how radiation therapy should be used to treat patients with basal cell carcinoma and cutaneous squamous cell carcinoma.
Recommendations defined when and how radiation therapy should be used to treat patients with basal cell carcinoma and cutaneous squamous cell carcinoma.

The submission to the FDA for the investigational CAR T-cell therapy is based on data from the phase II ZUMA-II trial for the treatment of adult patients with relapsed/refractory mantle cell lymphoma.

The drug candidate tipifarnib (Zarnestra) is being clinically studied for the treatment of patients with HRAS-mutant head and neck squamous cell carcinoma after progression on platinum therapy.
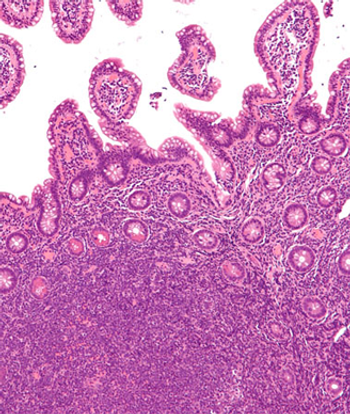
The ZUMA-II trial suggested that patients with relapsed/refractory mantle cell lymphoma resistant to prior therapies may benefit from the autologous anti-CD19 CAR T-cell therapy.





















