It was noted that the CA19-9 reductions in the 5 patients observed after infusion of satri-cel ranged from 51.3% to 96.1%.

It was noted that the CA19-9 reductions in the 5 patients observed after infusion of satri-cel ranged from 51.3% to 96.1%.

CARVac RNA vaccine may amplify the persistence of the BNT211 CAR T-cell therapy.
Schlechter discussed the potential advantages of TAC01-HER2 over current standards of care.
Schlechter discussed the design of the TACTIC-2 clinical trial for patients with HER2-positive solid tumors.
The chief medical officer of Triumvira Immunologics discussed the company’s future plans.

The chief medical officer of Triumvira Immunologics discussed how TAC01-HER2 could address unmet needs in treating solid tumors.

The deputy director of the Masonic Cancer Center discussed trends of presentations from ESMO 2022.

The chief medical officer of Triumvira Immunologics discussed updates from the TACTIC-2 study presented at ESMO Congress 2022.

The deputy director of the Masonic Cancer Center discussed the preclinical findings of his study on a TriKE engager and NK cells.

Hong presented data from the SURPASS clinical trial at the 2022 European Society for Medical Oncology (ESMO) Congress.

Fang commented on a limitation of the clinical trial pointed out at the European Society for Medical Oncology (ESMO) 2022 Congress.

Fang recently presented at the European Society for Medical Oncology (ESMO) 2022 Congress.

MT026 had an overall response rate of 83.3% in a first-in-human, investigator-initiated trial.
The FDA authorized the natural killer cell therapy combo for compassionate use.
Improved functional activity compared to ROR-1-targeted CAR-T therapies without the 2 forms of reprogramming was demonstrated in preclinical studies.
The 5-year relapse-free survival rate was 22.3% with T-VEC prior to surgery, compared with 15.2% for surgery alone.
Of the 8 treated patients who were evaluable, 6 patients had stable disease at Day 28.
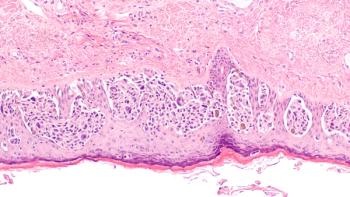
Based on the findings, investigators are considering registration with the FDA and EMA.
ADP-A2M4CD8 will be evaluated in the phase 2 SURPASS-3 trial initiating in late 2022 or early 2023.
Among the 31 patients evaluable for efficacy analysis, ORR was 100%.
A phase 2 trial evaluated the therapy in patients with diffuse large B-cell lymphoma, B-acute lymphocytic leukemia, and follicular lymphoma.
BNT211 showed encouraging responses in testicular cancer but less encouraging responses in ovarian cancer.
Thorsten Graef, MD, PhD, chief medical officer, Acepodia, discussed data on ACE1702 presented at ESMO 2021.