The placebo crossover cohort experienced statistically significant improvements in NSAA score over external control.

The placebo crossover cohort experienced statistically significant improvements in NSAA score over external control.
The reprioritization comes as the SIG-001 program remains on clinical hold after scarred and inviable cell spheres were observed in a treated patient.

Reductions in annualized bleeding rate remained significant at 2 years in the phase 3 study although recently presented phase 2 data showed significant FVIII reductions at 5 years.

AVRIOBIO announced that it is shifting priorities to focus on their gene therapy programs in Gaucher disease.

The DSMB has recommended that enrollment expand and doses be escalated from 3.3 to 5 mg in 1 cohort of the KIK-AS study.

The phase 3 GEM-3 study met both its primary and secondary end points compared with placebo in dEB.
Poseida’s announcement follows their presentation of positive data in metastatic castrate-resistant prostate cancer in September 2021.
The program is still on clinical hold following SUSARs of myelodysplastic syndrome in treated participants.
Annualized bleeding rate, spontaneous bleeding, and joint bleeding rates were all reduced in patients dosed with fitusiran compared to those receiving on-demand treatment.
Over half of patients in the fitusiran arm of the ATLAS-INH study had 0 treated bleeding events.
Patients treated with the CAR T-cell therapy had an over 4-fold increase in event-free survival.
All evaluable treated participants experienced a reduction in vaso-occlusive events after treatment with ARU-1801.
CSL Behring plans to submit regulatory applications in the US and EU in the first half of 2022 for the AAV gene therapy etranacogene dezaparvovec.

Cedrik Britten, MD, chief medical officer, Immatics discussed data on IMA203 presented at SITC 2021.
Adicet Bio reported positive interim data from the phase 1 GLEAN study.
An independent Data Safety and Monitoring Board has recommended that the phase 1 RePAIR trial continue at an accelerated pace.

Beam Therapeutics will assess the safety and efficacy of BEAM-101 for SCD in the phase 1/2 BEACON-101 study.
CYNK-101 is set to be evaluated in combination with standard chemotherapy, trastuzumab and pembrolizumab in a phase 1/2a study.
CRISPR Therapeutics presented positive data on 2 preclinical programs at the 2021 SITC meeting.
Pfizer and Sangamo voluntarily paused the trial after participants experienced FVIII activity levels of over 150%.
The investigational gene therapy, which will receive priority review, has a PDUFA date of May 20, 2022.

A 1-stage factor VIII assay revealed no apparent decrease in factor VIII activity over time.
The gene-edited approach hopes to overcome with the shortcomings of autologous hematopoietic stem-cell transplantation.

Following updated safety data, Rocket Pharmaceuticals has decided to discontinue the high-dose cohort.
The phase 1 trial is the first in-human trial to assess a gene therapy for CMT.
The Doheny Image Reading and Research Lab is leading a sub study to analyze OCT images with their proprietary 3D-OCTOR software.
The NK cell therapy is also being evaluated in a phase 1 study for acute myeloid leukemia.
Overall survival was 72% at 12 months but dropped to 54% at 14.6 months.
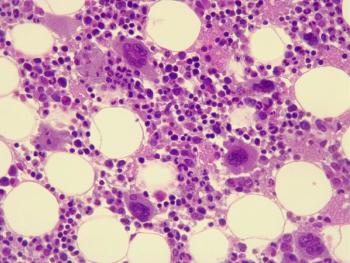
The request aims to ensure the comparability of omidubicel manufactured at different sites.
The first in-human study of CAR macrophages has dosed 2 participants so far.