
According to Novartis, it is the first gene replacement therapy to have been approved “for this broad population.”

According to Novartis, it is the first gene replacement therapy to have been approved “for this broad population.”

The NSGC president-elect spoke on the evolving role of genetic counselors, emphasizing personalized care, gene therapy advancements, and the need for multidisciplinary integration in healthcare.

Review top news and interview highlights from the week ending November 21, 2025.

The genetic counselor in the Department of Ophthalmology at the University of Pittsburgh Medical Center discussed the growing role of genetic counselors in guiding patients undergoing gene therapy.

Deborah Phippard, PhD, and Renier Brentjens, MD, PhD, spoke about strategies for streamlining development of novel therapies.

Catch up on the latest news, breakthroughs, and announcements from biotechnology companies making advancements in cell and gene therapies.
This finding came from an analysis of 37 patients treated in the phase 1/2 LEGEND trial.

Deborah Phippard, PhD, and Renier Brentjens, MD, PhD, discussed major friction points in cell and gene therapy development and uptake in the current day.

The chair of the Division of Hematology at Mayo Clinic discussed practical considerations for the introduction of cell therapies for the treatment of hematologic malignancies.

The vice president of Rare Disease Strategy at Worldwide Clinical Trials discussed the increasing importance of genetic counselors in clinical trials with regard to design, execution, and interpretation.

The president of the National Society of Genetic Counselors discussed AI and gene therapy, which constituted key topics at NSGC's annual conference this year.

The senior director of research and real world data at Genome Medical discussed multiple perspectives on how AI may take over some of the roles of genetic counselors in the future.

The senior director of research and real world data at Genome Medical discussed important considerations for genetic counselors thinking about using AI tools in their practice.

The senior director of research and real world data at Genome Medical discussed a session she chaired at the NSGC Annual Conference.
According to Intellia, the FDA placed clinical holds on both MAGNITUDE and MAGNITUDE-2 on October 29, 2025.

Review top news and interview highlights from the week ending November 7, 2025.

Deborah Phippard, PhD, and Renier Brentjens, MD, PhD, discussed major events in the past quarter century of cell and gene therapy research.

Catch up on the latest news, breakthroughs, and announcements from biotechnology companies making advancements in cell and gene therapies.

Naji Gehchan, MD, MBA, the chief medical and development officer of Kyverna Therapeutics, went over new data the company presented at AANEM’s 2025 meeting.
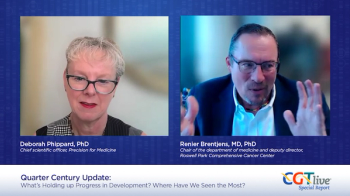
Deborah Phippard, PhD, and Renier Brentjens, MD, PhD, discussed how AI will affect cell and gene therapy research and practice.
uniQure pointed out that this new feedback stands in contrast to its previous communications with the FDA in several Type B meetings.

Catch up on any of the key data updates you may have missed last month, with coverage highlights from the CGTLive® team.

Catch up on any of the key FDA news stories you may have missed last month, with coverage highlights from the CGTLive® team.

Review top news and interview highlights from the week ending October 31, 2025.
Deborah Phippard, PhD, shared her thoughts with Renier Brentjens, MD, PhD, on reducing costs for cell and gene therapy products.

The chief medical and development officer of Kyverna Therapeutics went over new data the company presented at AANEM’s 2025 meeting.
Deborah Phippard, PhD, and Renier Brentjens, MD, PhD, shared their thoughts on the future of cell and gene therapy.
Deborah Phippard, PhD, shared her thoughts with Renier Brentjens, MD, PhD, about the importance of big wins for cell and gene therapy research.

Catch up on the latest news, breakthroughs, and announcements from biotechnology companies making advancements in cell and gene therapies.
Deborah Phippard, PhD, and Renier Brentjens, MD, PhD, shared advice for young scientists entering the cell and gene therapy field.