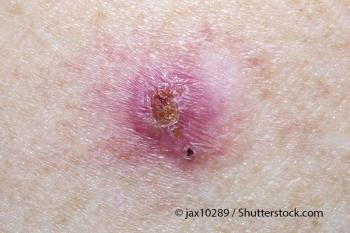
Researchers evaluated the response of patients with BCC to fractionated photodynamic therapy at 30 days after treatment.

Researchers evaluated the response of patients with BCC to fractionated photodynamic therapy at 30 days after treatment.
Researchers tested the addition of the T cell–boosting decitabine to anti–PD-1 therapy with camrelizumab among patients with relapsed or refractory classic Hodgkin lymphoma.

Christine N. Duncan, MD, discusses the current components of CAR T-cell therapy in pediatric acute lymphoblastic leukemia.
Study results demonstrated that in adult patients with blastic plasmacytoid dendritic cell neoplasm, the targeted therapy resulted in high response rates, particularly among treatment-naïve patients.

The combination of lenalidomide (Revlimid) with rituximab (Rituxan), cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP; R2-CHOP) did not improve progression-free survival compared with placebo and R-CHOP as a frontline therapy in patients with activated B-cell-type diffuse large B-cell lymphoma, missing the primary endpoint of the phase III ROBUST trial.

Tagraxofusp led to a 90% overall response rate as a first-line therapy in patients with blastic plasmacytoid dendritic cell neoplasm.
Modifications to the engineering of the chimeric antigen receptor (CAR) mean that the patient produces fewer cytokines and has time to clear them before they build up in the bloodstream.
As part of CMS’ FY 2020 Medicare Hospital Inpatient Prospective Payment System and Long-Term Acute Care Hospital Prospective Payment System Proposed Rule and Request for Information, the agency is proposing an increase in how much it reimburses hospitals for chimeric antigen receptor (CAR) T-cell therapy, as well as wage index hikes for rural hospitals.

Omar Nadeem, MD, highlights exciting approaches that have emerged in relapsed/refractory myeloma, such as CAR T-cell therapy, and shares unanswered questions that future research should address.
Phase 2 trial results suggest the possibility of a treatment that may regenerate brain cells following TBI.
Dozens of doctors and other medical professionals have been charged with exchanging opioids and other drugs for sex and cash; the first 2 patients in the United States have been treated with CRISPR; doctors have cured infants of immunodeficiency syndrome using gene therapy made from HIV.

A phase III trial evaluated intermediate-dose cytarabine plus granulocyte-colony stimulating factor (G-CSF) vs G-CSF alone prior to autoSCT in multiple myeloma.

Academic medical centers and a group representing community oncology practices have both raised concerns about CMS’ proposed reimbursement plan for chimeric antigen receptor (CAR) T-cell therapy, the individually manufactured gene treatments that are revolutionizing cancer care. The plan will be finalized next month, a year after the federal government launched a national coverage analysis to determine how to pay for these lifesaving yet expensive cancer treatments.

Patients with hematologic malignancy who are undergoing chemotherapy or a conditioning regimen for hematopoietic stem cell transplant (HSCT) are at high risk of infection because of the severity and duration of neutropenia. Fever with neutropenia is a common presentation that suggests an infection leading to empiric antibacterial therapy. To prevent infection and thus the neutropenic fever, antibacterial prophylaxis, especially with fluoroquinolones, emerged as a common practice based on results of 2 randomized controlled trials published in 2005 that showed reduced incidence of fever and bacteremia despite lack of a mortality benefit.

In this article, important concepts in the molecular testing of non–small-cell lung cancer are highlighted.

No overall survival advantage was obtained from maintenance therapy comprising nivolumab (Opdivo) alone or in combination with ipilimumab (Yervoy) over placebo in patients with extensive-stage small cell lung cancer.

Prasad S. Adusumilli, MD, sheds light on the preliminary phase I data from a trial of CAR T-cell therapy in malignant pleural disease from mesothelioma.
Research presented at AACR 2019 evaluated autologous mesothelin-targeted chimeric antigen receptor T-cell therapy in patients with malignant pleural disease.
A phase I trial evaluated the safety and efficacy of adding lymphodepletion to HER2-targeted T-cell therapy in patients with advanced HER2-positive sarcoma.

The planned phase 1/2 trial of the recombinant AAV5 vector treatment, the first one-time administered AAV gene therapy to enter clinical testing for Huntington disease, is expected to begin dosing patients in the second half of 2019.

Researchers have identified a potential new target for CAR T-cell therapy in patients with multiple myeloma.

Tumor mutational burden identified patients who obtained a survival benefit with the PD-L1 inhibitor durvalumab, as initial therapy versus chemotherapy for advanced non–small cell lung cancer, even though the primary analysis of the randomized trial showed no difference between treatment groups, according to a new analysis.

Patients with multiple myeloma who relapsed following allogeneic stem cell transplantation and also failed several posttransplant therapies demonstrated a strong response to daratumumab salvage therapy.

CAR T cells targeting mesothelin-expressing tumors demonstrated safety and efficacy in a preliminary clinical evaluation in patients with malignant pleural disease.

The administration of HER2-directed CAR T-cell therapy and lymphodepletion chemotherapy demonstrated antitumor activity and was found to be safe in pediatric and adult patients with advanced HER2-positive sarcoma.

Although CAR T-cell therapy has not yet proved effective against solid tumors, a novel CAR that targets mesothelin proteins expressed in pancreatic cancer is showing early signs of activity.
Recent work looking at the response to immune checkpoint blockade (ICB) therapy given before surgery for advanced melanoma found increased infiltration of B cells, a type of white blood cell, in patients who responded to therapy compared with those who did not.

Sequential administration of CAR T-cell therapy targeting both the CD19 and CD22 antigens demonstrated high complete remission rates and improved long-term survival in patients with relapsed/refractory B-cell acute lymphoblastic leukemia following allogeneic hematopoietic stem cell transplant.

Lazaros J. Lekakis, MD, discusses the promise of CAR T cells and some unanswered questions with this rapidly emerging therapy.

Experts shared the challenges they've encountered in integrating novel treatments such as CAR T-cell therapy into practice at the 2019 NCCN Annual Conference.