
Arnaud Lacoste, PhD, the chief scientific officer of Aurion Biotech, discussed AURN001, a corneal endothelial cell therapy that was recently approved in Japan, and had a nonclinical data read out at ARVO 2023.

Arnaud Lacoste, PhD, the chief scientific officer of Aurion Biotech, discussed AURN001, a corneal endothelial cell therapy that was recently approved in Japan, and had a nonclinical data read out at ARVO 2023.

The head of the Department of Ophthalmology at the Center for Medical Genetics at Ghent University Hospital discussed the need to reevaluate requirements for regulatory approval for ophthalmology gene therapies.

Krystal Biotech's B-VEC was well-tolerated and the treated patient experienced significant improvement in visual acuity.

Review top news and interview highlights from the week ending May 5, 2023.

Catch up on the latest news, breakthroughs, and announcements from biotechnology companies making advancements in cell and gene therapies.

The head of the Department of Ophthalmology at the Center for Medical Genetics, Ghent University Hospital, discussed the main takeaways of a post-marketing study on the gene therapy.

Atsena anticipates initiating the Lighthouse study of ATSN-201 in mid-2023.

CGTLive's sister publication spoke with Barth, the chief medical officer at Ascidian, about the company’s attempts to develop a new method of administration for gene therapy at ARVO 2023.
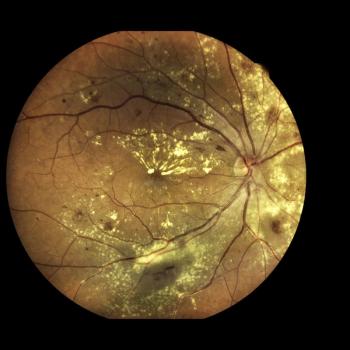
New data from imaging analyses on RG6501 were presented at the ARVO 2023 Annual Meeting.

The study found significant improvements in BCVA, in contrast to several other studies evaluating Luxturna.

The data, presented at ARVO's 2023 conference, also showed a favorable safety profile.

Review top news and interview highlights from the week ending April 28, 2023.
The chief scientific officer of Aurion Biotech discussed the properties of the cell therapy for corneal dystrophies, which was recently approved in Japan.


The new data builds upon positive long-term follow-up data from clinical trials.

The head of the Department of Ophthalmology at the Center for Medical Genetics, Ghent University Hospital, discussed the results of a post-marketing study presented at ARVO’s 2023 conference.

The safety profile of lenadogene nolparvovec at 5-years post treatment was similar to 2-year data.

Catch up on the latest news, breakthroughs, and announcements from biotechnology companies making advancements in cell and gene therapies.

No serious TEAEs have been related to ATSN-101 with data up to 6 months after treatment.
The chief scientific officer of Aurion Biotech discussed the investigational cell therapy AURN001.

STZ eyetrial’s treatment rAAV8.hPDE6A was otherwise deemed well-tolerated.

The commercially available gene therapy, developed by Spark Therapeutics, was evaluated in multiple studies presented at ARVO’s 2023 conference.
The new positive data on the AAV based gene therapy builds upon real-world data presented at ARVO’s conference in 2022.

The investigators from Children’s Hospital of Philadelphia discussed the follow-up studies they are conducting in light of their recent findings regarding DNA virus replication.

Catch up on the latest news, breakthroughs, and announcements from biotechnology companies making advancements in cell and gene therapies.