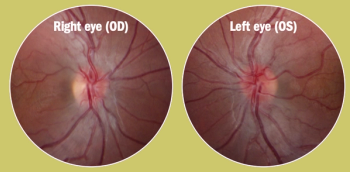
Neurophth Therapeutics, Inc (Neurophth) and AAVnerGene Inc have announced the launch of a strategic partnership that will grant Neurophth global rights to mutually select adeno-associated virus (AAV) capsids for the creation of the next-generation ophthalmic gene therapy.